Genital wart
| Genital warts | |
|---|---|
| Other names | Condylomata acuminata, venereal warts, anal warts, anogenital warts |
| Frequency | ~1% (US)[4] |
Genital warts are a
HPV types 6 and 11 are responsible for causing majority of genital warts whereas HPV types 16, 18, 31, 33, and 35 are also occasionally found.
Some
About 1% of people in the United States have genital warts.[4] Many people, however, are infected and do not have symptoms.[4] Without vaccination nearly all sexually active people will get some type of HPV at one point in their lives.[9][10] The disease has been known at least since the time of Hippocrates in 300 BC.[11]
Signs and symptoms

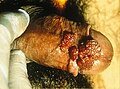

They may be found anywhere in the anal or genital area, and are frequently found on external surfaces of the body, including the
They can be as small as 1–5 mm in diameter, but can also grow or spread into large masses in the genital or anal area. In some cases they look like small stalks. They may be hard ("keratinized") or soft. Their color can be variable, and sometimes they may bleed.[13]
In most cases, there are no symptoms of HPV infection other than the warts themselves. Sometimes warts may cause itching, redness, or discomfort, especially when they occur around the anus. Although they are usually without other physical symptoms, an outbreak of genital warts may cause psychological distress, such as anxiety, in some people.[14]
Causes
Transmission
HPV is most commonly transmitted through
The types of HPV that cause warts are highly transmissible. Roughly three out of four unaffected partners of patients with warts develop them within eight months.[16] Other studies of partner concordance suggest that the presence of visible warts may be an indicator of increased infectivity; HPV concordance rates are higher in couples where one partner has visible warts.[15]
Latency and recurrence
Although 90% of HPV infections are cleared by the body within two years of infection, it is possible for infected cells to undergo a latency (quiet) period, with the first occurrence or a recurrence of symptoms happening months or years later.[4] Latent HPV, even with no outward symptoms, is still transmissible to a sexual partner. If an individual has unprotected sex with an infected partner, there is a 70% chance that he or she will also become infected.[citation needed]
In individuals with a history of previous HPV infection, the appearance of new warts may be either from a new exposure to HPV, or from a recurrence of the previous infection. As many as one-third of people with warts will experience a recurrence.[17]
Children
Anal or genital warts may be transmitted during birth. The presence of wart-like lesions on the genitals of young children has been suggested as an indicator of sexual abuse. However, genital warts can sometimes result from autoinoculation by warts elsewhere on the body, such as from the hands.[18] It has also been reported from sharing of swimsuits, underwear, or bath towels, and from non-sexual touching during routine care such as diapering. Genital warts in children are less likely to be caused by HPV subtypes 6 and 11 than adults, and more likely to be caused by HPV types that cause warts elsewhere on the body ("cutaneous types"). Surveys of pediatricians who are child abuse specialists suggest that in children younger than 4 years old, there is no consensus on whether the appearance of new anal or genital warts, by itself, can be considered an indicator of sexual abuse.[19]
Diagnosis

The diagnosis of genital warts is most often made visually, but may require confirmation by biopsy in some cases.[20] Smaller warts may occasionally be confused with molluscum contagiosum.[19] Genital warts,
Some practitioners use an
Prevention
In the UK, Gardasil replaced Cervarix in September 2012[22] for reasons unrelated to safety.[23] Cervarix had been used routinely in young females from its introduction in 2008, but was only effective against the high-risk HPV types 16 and 18, neither of which typically causes warts.[citation needed]
Management
There is no cure for HPV. Existing treatments are directed towards the removal of visible warts, but these may also regress on their own without any therapy.[14] There is no evidence to suggest that removing visible warts reduces transmission of the underlying HPV infection. As many as 80% of people with HPV will clear the infection within 18 months.[16]
A healthcare practitioner may offer one of several ways to treat warts, depending on their number, sizes, locations, or other factors. All treatments can cause depigmentation, itching, pain, or scarring.[14][24]
Treatments can be classified as either physically ablative, or topical agents. Physically ablative therapies are considered more effective at initial wart removal, but like all therapies have significant recurrence rates.[14][18]
Many therapies, including
Physical ablation
Physically ablative methods are more likely to be effective on keratinized warts. They are also most appropriate for patients with fewer numbers of relatively smaller warts.[18]
- Simple excision, such as with scissors under local anesthesia, is highly effective.[14]
- Liquid nitrogen cryosurgery is usually performed in an office visit, at weekly intervals. It is effective, inexpensive, safe for pregnancy, and does not usually cause scarring.[14]
- Electrocauterization (sometimes called "loop electrical excision procedure" or LEEP) is a procedure with a long history of use and is considered effective.[14]
- Laser ablation has less evidence to suggest its use. It may be less effective than other ablative methods.[18] It is extremely expensive, and often used as a last resort.[26]
- Formal surgical procedures, performed by a
Topical agents
- A 0.15–0.5% podophyllin (see below). Podofilox is safer and more effective than podophyllin.[27] Skin erosion and pain are more commonly reported than with imiquimod and sinecatechins.[28] Its use is cycled (two times per day for 3 days then 4–7 days off); one review states that it should only be used for four cycles.[27]
- Imiquimod is a topical immune response cream, applied to the affected area. It causes less local irritation than podofilox but may cause fungal infections (11% in package insert) and flu-like symptoms (less than 5% disclosed in package insert).[28] It does not significantly impact the occurrence of new warts or the systemic reactions.[29]
- Sinecatechins is an ointment of catechins (55% epigallocatechin gallate[26]) extracted from green tea and other components. Mode of action is undetermined.[30] It appears to have higher clearance rates than podophyllotoxin and imiquimod and causes less local irritation, but clearance takes longer than with imiquimod.[28]
- Trichloroacetic acid (TCA) is less effective than cryosurgery,[27] and is not recommended for use in the vagina, cervix, or urinary meatus.[26]
- Interferon can be used; it is effective, but it is also expensive and its effect is inconsistent.[27]
Discontinued
- A 5% 5-fluorouracil (5-FU) cream was used, but it is no longer considered an acceptable treatment due to the side-effects.[26]
Podophyllin, podofilox and isotretinoin should not be used during pregnancy, as they could cause birth defects in the fetus.
Oral agents
Isotretinoin taken orally has been shown to treat recalcitrant condylomata acuminata (RCA) of the cervix.
Epidemiology
Genital HPV infections have an estimated prevalence in the US of 10–20% and clinical manifestations in 1% of the sexually active adult population.[27] US incidence of HPV infection has increased between 1975 and 2006.[27] About 80% of those infected are between the ages of 17–33.[27] Although treatments can remove warts, they do not remove the HPV, so warts can recur after treatment (about 50–73% of the time[31]). Warts can also spontaneously regress (with or without treatment).[27]
Traditional theories postulated that the virus remained in the body for a lifetime. However, studies using sensitive DNA techniques have shown that through
Etymology
A condyloma acuminatum is a single genital wart, and condylomata acuminata are multiple genital warts. The word roots mean 'pointed wart' (from Greek κόνδυλος 'knuckle', Greek -ωμα -oma 'disease', and Latin acuminatum 'pointed'). Although similarly named, it is not the same as condyloma latum, which is a complication of secondary syphilis.[citation needed]
References
- ^ ISBN 978-0-323-54753-6.
- ^ ISBN 978-0-323-98644-1.
- ^ ISBN 9780323529570.
- ^ PMID 21121531.
- ^ a b c d e f g h i "CDC - Genital Warts - 2010 STD Treatment Guidelines". www.cdc.gov. 28 January 2011. Archived from the original on 8 July 2018. Retrieved 2 January 2018.
- ^ a b "Genital warts". NHS. 21 August 2017. Retrieved 2 January 2018.
- ISBN 978-0-323-55087-1.
- ISBN 978-1-35118-825-8. Retrieved July 11, 2023.
- ^ a b US National Cancer Institute. "HPV and Cancer". Retrieved 2 January 2018.
- ^ US Centers for Disease Control. "Genital HPV Infection - Fact Sheet". Retrieved 16 November 2017.
- ISBN 9780471971689.
- ^ Scheinfeld, Noah (2017-01-04). "Condylomata acuminata (anogenital warts) in adults: Epidemiology, pathogenesis, clinical features, and diagnosis". UpToDate. Retrieved 2018-01-01.
- ^ "Condylomata Acuminata (Genital Warts)". The Lecturio Medical Concept Library. Retrieved 10 July 2021.
- ^ PMID 22409368.
- ^ PMID 21075056.
- ^ PMID 22583720.
- PMID 22131115.
- ^ a b c d e "United Kingdom National Guideline on the Management of Anogenital Warts, 2007" (PDF). British Association for Sexual Health and HIV. Archived from the original (PDF) on 23 September 2015. Retrieved 3 August 2013.
- ^ S2CID 207170959.
- ^ Workowski, K; Berman, S. Sexually Transmitted Diseases Treatment Guidelines, 2010 (PDF). United States Centers for Disease Control. p. 70.
- ^ United States Food and Drug Administration. "Gardasil". Approved Products. Retrieved 1 January 2013.
- ^ UK Department of Health. "Your guide to the HPV vaccination from September 2012". Retrieved 1 January 2013.
- ^ UK Medicines and Healthcare products Regulatory Agency. "Human papillomavirus vaccine Cervarix: safety review shows balance of risks and benefits remains clearly positive". Archived from the original on 3 April 2013. Retrieved 1 January 2013.
- ^ PMID 15617297. Archived from the originalon 2008-07-20. Retrieved 2009-05-30.
- PMID 17210977.
- ^ S2CID 33302840.
- ^ PMID 16638419.
- ^ S2CID 31437162.
- PMID 25362229.
- ^ "Veregen label information" (PDF). Retrieved 2013-01-01.
- ^ CDC. (2004). REPORT TO CONGRESS: Prevention of Genital Human Papillomavirus Infection.
External links
- Human Papilloma Virus at Curlie
