Tin
 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tin | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Allotropes | silvery-white, β (beta); gray, α (alpha) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(Sn) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tin in the periodic table | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
kJ/mol | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | white (β): 296.1 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | white (β): 27.112 J/(mol·K) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
protohistoric, around 35th century BC | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symbol | "Sn": from Latin stannum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of tin | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Tin is a
Tin is a post-transition metal in group 14 of the periodic table of elements. It is obtained chiefly from the mineral cassiterite, which contains stannic oxide, SnO
2. Tin shows a chemical similarity to both of its neighbors in group 14, germanium and lead, and has two main oxidation states, +2 and the slightly more stable +4. Tin is the 49th-most abundant element on Earth and has, with 10 stable isotopes, the largest number of stable isotopes in the periodic table, due to its magic number of protons.
It has two main allotropes: at room temperature, the stable allotrope is β-tin, a silvery-white, malleable metal; at low temperatures it is less dense grey α-tin, which has the diamond cubic structure. Metallic tin does not easily oxidize in air and water.
The first tin alloy used on a large scale was bronze, made of 1⁄8 tin and 7⁄8 copper (12.5% and 87.5% respectively), from as early as 3000 BC. After 600 BC, pure metallic tin was produced. Pewter, which is an alloy of 85–90% tin with the remainder commonly consisting of copper, antimony, bismuth, and sometimes lead and silver, has been used for flatware since the Bronze Age. In modern times, tin is used in many alloys, most notably tin-lead soft solders, which are typically 60% or more tin, and in the manufacture of transparent, electrically conducting films of indium tin oxide in optoelectronic applications. Another large application is corrosion-resistant tin plating of steel. Because of the low toxicity of inorganic tin, tin-plated steel is widely used for food packaging as "tin cans". Some organotin compounds can be extremely toxic.
Characteristics
Physical

Tin is a soft,
| External videos | |
|---|---|
β-tin, also called white tin, is the
In cold conditions β-tin tends to transform spontaneously into α-tin, a phenomenon known as "tin pest" or "tin disease".[15] Some unverifiable sources also say that, during Napoleon's Russian campaign of 1812, the temperatures became so cold that the tin buttons on the soldiers' uniforms disintegrated over time, contributing to the defeat of the Grande Armée,[16] a persistent legend.[17][18][19]
The α-β transformation temperature is 13.2 °C (55.8 °F), but impurities (e.g. Al, Zn, etc.) lower it well below 0 °C (32 °F). With the addition of antimony or bismuth the transformation might not occur at all, increasing durability.[20]
Commercial grades of tin (99.8% tin content) resist transformation because of the inhibiting effect of small amounts of bismuth, antimony, lead, and silver present as impurities. Alloying elements such as copper, antimony, bismuth, cadmium, and silver increase the hardness of tin.
Tin becomes a
Chemical
Tin resists corrosion from
Isotopes
Tin has ten
Tin is one of the easiest elements to detect and analyze by
4).[b][26]
Of the stable isotopes, tin-115 has a high capture cross section for fast neutron energies, at 30 barns. Tin-117 ranks one below, with a cross section of 2.3 barns, while tin-119 has a slightly smaller cross section of 2.2 barns. [27] Before these cross sections were well known, it was proposed to use tin-lead solder as a reactor coolant for fast reactors because of its low melting point. Current studies are for lead or lead-bismuth reactor coolants because both heavy metals are nearly transparent to fast neutrons, with very low capture cross sections. [28] In order to use a tin or tin-lead coolant the tin would first have to go through isotopes separation to remove the 115, 117 and 119 tin isotopes. Combined, these three isotopes make up about 17% of natural tin but represent nearly all of the capture cross section. Of the remaining seven isotopes tin-112 has a capture cross section of 1 barn. The other six isotopes forming 82.7% of natural tin have capture cross sections of 0.3 barns or less making them effectively transparent to neutrons, like lead and bismuth are.
Tin has 31 unstable isotopes, ranging in mass number from 99 to 139. The unstable tin isotopes have half-lives of less than a year except for
The relative differences in the number of tin's stable isotopes can be explained by how they are formed during
Etymology
The word tin is shared among
The
The Meyers Konversations-Lexikon suggests instead that stannum came from Cornish stean, and is evidence that Cornwall in the first centuries AD was the main source of tin.[citation needed]
History

Tin extraction and use can be dated to the beginnings of the Bronze Age around 3000 BC, when it was observed that
Compounds and chemistry
In the great majority of its compounds, tin has the
Inorganic compounds
Halide compounds are known for both oxidation states. For Sn(IV), all four halides are well known: SnF4, SnCl4, SnBr4, and SnI4. The three heavier members are volatile molecular compounds, whereas the tetrafluoride is polymeric. All four halides are known for Sn(II) also: SnF2, SnCl
2, SnBr2, and SnI2. All are polymeric solids. Of these eight compounds, only the iodides are colored.[41]
Tin(II) chloride (also known as stannous chloride) is the most important commercial tin halide. Illustrating the routes to such compounds, chlorine reacts with tin metal to give SnCl4 whereas the reaction of hydrochloric acid and tin produces SnCl
2 and hydrogen gas. Alternatively SnCl4 and Sn combine to stannous chloride by a process called comproportionation:[42]
- SnCl4 + Sn → 2 SnCl
2
Tin can form many oxides, sulfides, and other
6]2−, like K
2[Sn(OH)
6], are also known, though the free stannic acid H
2[Sn(OH)
6] is unknown.
Sulfides of tin exist in both the +2 and +4 oxidation states: tin(II) sulfide and tin(IV) sulfide (mosaic gold).
2)[44]
Hydrides
Stannane (SnH
4), with tin in the +4 oxidation state, is unstable. Organotin hydrides are however well known, e.g. tributyltin hydride (Sn(C4H9)3H).[11] These compound release transient tributyl tin radicals, which are rare examples of compounds of tin(III).[45]
Organotin compounds
Most organotin compounds are colorless liquids or solids that are stable to air and water. They adopt tetrahedral geometry. Tetraalkyl- and tetraaryltin compounds can be prepared using Grignard reagents:[47]
- SnCl
4 + 4 RMgBr → R
4Sn + 4 MgBrCl
The mixed halide-alkyls, which are more common and more important commercially than the tetraorgano derivatives, are prepared by
- SnCl
4 + R
4Sn → 2 SnCl
2R2
Divalent organotin compounds are uncommon, although more common than related divalent
Occurrence

Tin is generated via the long s-process in low-to-medium mass stars (with masses of 0.6 to 10 times that of the Sun), and finally by beta decay of the heavy isotopes of indium.[50]
Tin is the 49th most abundant element in
Tin does not occur as the native element but must be extracted from various ores. Cassiterite (SnO
2) is the only commercially important source of tin, although small quantities of tin are recovered from complex sulfides such as stannite, cylindrite, franckeite, canfieldite, and teallite. Minerals with tin are almost always associated with granite rock, usually at a level of 1% tin oxide content.[52]
Because of the higher
| Country | Reserves |
|---|---|
| 1,500,000 | |
| 250,000 | |
| 310,000 | |
| 800,000 | |
| 590,000 | |
| 400,000 | |
| 350,000 | |
| 180,000 | |
| 170,000 | |
| Other | 180,000 |
| Total | 4,800,000 |
| Year | Million tonnes |
|---|---|
| 1965 | 4,265 |
| 1970 | 3,930 |
| 1975 | 9,060 |
| 1980 | 9,100 |
| 1985 | 3,060 |
| 1990 | 7,100 |
| 2000 | 7,100[54] |
| 2010 | 5,200[54] |
About 253,000 tonnes of tin were mined in 2011, mostly in China (110,000 t), Indonesia (51,000 t), Peru (34,600 t), Bolivia (20,700 t) and Brazil (12,000 t).[54] Estimates of tin production have historically varied with the market and mining technology. It is estimated that, at current consumption rates and technologies, the Earth will run out of mine-able tin in 40 years.[55] In 2006 Lester Brown suggested tin could run out within 20 years based on conservative estimates of 2% annual growth.[56]
Scrap tin is an important source of the metal. Recovery of tin through recycling is increasing rapidly.[when?][citation needed] Whereas the United States has neither mined (since 1993) nor smelted (since 1989) tin, it was the largest secondary producer, recycling nearly 14,000 tonnes in 2006.[54]
New deposits are reported in Mongolia,[57] and in 2009, new deposits of tin were discovered in Colombia.[58]
Production
Tin is produced by
- SnO2 + C Sn + CO2↑
Mining and smelting
Industry
The ten largest companies produced most of the world's tin in 2007.
Most of the world's tin is traded on LME, from 8 countries, under 17 brands.[62]
| Company | Polity | 2006 | 2007 | 2017[64] | 2006–2017 % change |
|---|---|---|---|---|---|
| Yunnan Tin | China | 52,339 | 61,129 | 74,500 | 42.3 |
| PT Timah | Indonesia | 44,689 | 58,325 | 30,200 | −32.4 |
| Malaysia Smelting Corp | Malaysia | 22,850 | 25,471 | 27,200 | 19.0 |
| Yunnan Chengfeng | China | 21,765 | 18,000 | 26,800 | 23.1 |
| Minsur | Peru | 40,977 | 35,940 | 18,000 | −56.1 |
| EM Vinto | Bolivia | 11,804 | 9,448 | 12,600 | 6.7 |
| Guangxi China Tin | China | / | / | 11,500 | / |
| Thaisarco | Thailand | 27,828 | 19,826 | 10,600 | −61.9 |
| Metallo-Chimique | Belgium | 8,049 | 8,372 | 9,700 | 20.5 |
| Gejiu Zi Li | China | / | / | 8,700 | / |
International Tin Council was established in 1947 to control the price of tin. It collapsed in 1985. In 1984, Association of Tin Producing Countries was created, with Australia, Bolivia, Indonesia, Malaysia, Nigeria, Thailand, and Zaire as members.[65]
Price and exchanges
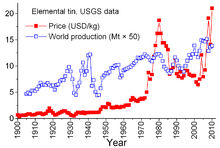
Tin is unique among mineral commodities because of the complex agreements between producer countries and consumer countries dating back to 1921. Earlier agreements tended to be somewhat informal and led to the "First International Tin Agreement" in 1956, the first of a series that effectively collapsed in 1985. Through these agreements, the International Tin Council (ITC) had a considerable effect on tin prices. ITC supported the price of tin during periods of low prices by buying tin for its buffer stockpile and was able to restrain the price during periods of high prices by selling from the stockpile. This was an anti-free-market approach, designed to assure a sufficient flow of tin to consumer countries and a profit for producer countries. However, the buffer stockpile was not sufficiently large, and during most of those 29 years tin prices rose, sometimes sharply, especially from 1973 through 1980 when rampant inflation plagued many world economies.[66]
During the late 1970s and early 1980s, the U.S. reduced its strategic tin stockpile, partly to take advantage of historically high tin prices. The

London Metal Exchange (LME) is tin's principal trading site.[54] Other tin contract markets are Kuala Lumpur Tin Market (KLTM) and Indonesia Tin Exchange (INATIN).[67]
Due to factors involved in the
Applications
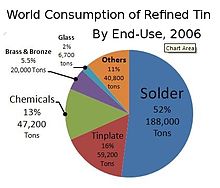
In 2018, just under half of all tin produced was used in solder. The rest was divided between tin plating, tin chemicals, brass and bronze alloys, and niche uses.[69]
Solder

Tin has long been used in alloys with lead as
Tin plating
Tin bonds readily to
Copper cooking vessels such as saucepans and frying pans are frequently lined with a thin plating of tin, by electroplating or by traditional chemical methods, since use of copper cookware with acidic foods can be toxic.
Specialized alloys


Tin in combination with other elements forms a wide variety of useful alloys. Tin is most commonly alloyed with copper.
The
A small percentage of tin is added to
Most metal pipes in a pipe organ are of a tin/lead alloy, with 50/50 as the most common composition. The proportion of tin in the pipe defines the pipe's tone, since tin has a desirable tonal resonance. When a tin/lead alloy cools, the lead phase solidifies first, then when the eutectic temperature is reached, the remaining liquid forms the layered tin/lead eutectic structure, which is shiny; contrast with the lead phase produces a mottled or spotted effect. This metal alloy is referred to as spotted metal. Major advantages of using tin for pipes include its appearance, workability, and resistance to corrosion.[82][83]
Optoelectronics
The
Other applications

Punched tin-plated steel, also called pierced tin, is an artisan technique originating in central Europe for creating functional and decorative housewares. Decorative piercing designs exist in a wide variety, based on local tradition and the artisan. Punched tin lanterns are the most common application of this artisan technique. The light of a candle shining through the pierced design creates a decorative light pattern in the room where it sits. Lanterns and other punched tin articles were created in the New World from the earliest European settlement. A well-known example is the Revere lantern, named after Paul Revere.[85]
Before the modern era, in some areas of the Alps, a goat or sheep's horn would be sharpened and a tin panel would be punched out using the alphabet and numbers from one to nine. This learning tool was known appropriately as a "tin horn". Modern reproductions are decorated with such motifs as hearts and tulips.
In America, pie safes and food safes were in use in the days before refrigeration. These were wooden cupboards of various styles and sizes – either floor standing or hanging cupboards meant to discourage vermin and insects and to keep dust from perishable foodstuffs. These cabinets had tinplate inserts in the doors and sometimes in the sides, punched out by the homeowner, cabinetmaker, or a tinsmith in varying designs to allow for air circulation while excluding flies. Modern reproductions of these articles remain popular in North America.[86]
Window glass is most often made by floating molten
Tin is used as a negative electrode in advanced
Tin is used as a target to create laser-induced plasmas that act as the light source for extreme ultraviolet lithography.
Organotin compounds
The organotin compounds are most heavily used. Worldwide industrial production probably exceeds 50,000 tonnes.[92]
PVC stabilizers
The major commercial application of organotin compounds is in the stabilization of
Biocides
Some organotin compounds are relatively toxic, with both advantages and problems. They are used for
Organic chemistry
Some tin reagents are useful in organic chemistry. In the largest application, stannous chloride is a common reducing agent for the conversion of nitro and oxime groups to amines. The Stille reaction couples organotin compounds with organic halides or pseudohalides.[104]
Li-ion batteries
Tin forms several inter-metallic phases with lithium metal, making it a potentially attractive material for battery applications. Large volumetric expansion of tin upon alloying with lithium and instability of the tin-organic electrolyte interface at low electrochemical potentials are the greatest challenges to employment in commercial cells.[105] Tin inter-metallic compound with cobalt and carbon was implemented by Sony in its Nexelion cells released in the late 2000s. The composition of the active material is approximately Sn0.3Co0.4C0.3. Research showed that only some crystalline facets of tetragonal (beta) Sn are responsible for undesirable electrochemical activity.[106]
Precautions
Cases of poisoning from tin metal, its oxides, and its salts are almost unknown. On the other hand, certain
Exposure to tin in the workplace can occur by inhalation, skin contact, and eye contact. The US
See also
- Cassiterides (the mythical Tin Islands)
- Stannary
- Terne
- Tin pest
- Tin mining in Britain
- Tinning
- Whisker (metallurgy) (tin whiskers)
Notes
- ^ The thermal expansion of β-Sn is anisotropic: the parameters (at 20 °C) for each crystal axis are αa = 16.19×10−6/K, αc = 32.89×10−6/K, and αaverage = αV/3 = 21.76×10−6/K.[3]
- ^ Only hydrogen, fluorine, phosphorus, thallium and xenon are easier to use NMR analysis with for samples containing isotopes at their natural abundance.
References
- ^ "Standard Atomic Weights: Tin". CIAAW. 1983.
- ISSN 1365-3075.
- ^ ISBN 978-1-62708-155-9.
- ^ "New Type of Zero-Valent Tin Compound". Chemistry Europe. 27 August 2016.
- ^ "HSn". NIST Chemistry WebBook. National Institute of Standards and Technology. Retrieved 2013-01-23.
- ^ "SnH3". NIST Chemistry WebBook. National Institure of Standards and Technology. Retrieved 2013-01-23.
- ISBN 0-8493-0486-5.
- ISBN 0-8493-0464-4.
- .
- ^ Gray, Theodore (2007). "Tin Images". The Elements. Black Dog & Leventhal.
- ^ ISBN 978-3-11-007511-3.
- ^ "Ink with tin nanoparticles could print future circuit boards". Phys.org. 12 April 2011. Archived from the original on 16 September 2011.
- S2CID 25202674.
- S2CID 120417927.
- ^ "Tin Pests | Center for Advanced Life Cycle Engineering". calce.umd.edu. Retrieved 2022-11-04.
- ^ Le Coureur, Penny; Burreson, Jay (2004). Napoleon's Buttons: 17 molecules that changed history. New York: Penguin Group, USA.
- ISBN 978-0-19-966109-1.
- ^ Cotton, Simon (29 April 2014). "Book review: The last alchemist in Pari". Chemistry World. Royal Society of Chemistry. Archived from the original on 10 August 2014. Retrieved 22 November 2019.
- ISBN 978-0-19-960563-7.
Only officers had metal buttons, and those were made of brass.
- ^ ISBN 978-1-56676-661-6.
- ^ "Tin Alloys – Characteristics and Uses". Nuclear Power. Retrieved 2022-11-04.
- .
- ^ S2CID 37842752.
- ISBN 978-0-87170-518-1. Archivedfrom the original on 2016-05-11.
- ^ Crutchlow, Charlotte (25 June 2021). "Tin-Formation About the Element Tin | Periodic Table". ChemTalk. Retrieved 2022-11-04.
- ^ "Interactive NMR Frequency Map". Archived from the original on 2011-06-04. Retrieved 2009-05-05.
- ^ "Neutron Scattering Lengths and cross sections".
- ^ "Westinghouse Nuclear > Energy Systems > Lead-cooled Fast Reactor".
- .
- S2CID 120201972. Archived from the original(PDF) on 2011-10-21.
- ^ a b c "tin". Oxford English Dictionary (Online ed.). Oxford University Press. (Subscription or participating institution membership required.)
- ^ Harper, Douglas. "tin". Online Etymology Dictionary.
- ^ Encyclopædia Britannica, 11th Edition, 1911, s.v. 'tin', citing H. Kopp
- ^ "The Ancient Mining of Tin". oxleigh.freeserve.co.uk. Archived from the original on 2009-04-03. Retrieved 2009-07-07.
- American Heritage Dictionary
- ISBN 978-1-84171-564-3.
- ^ ISBN 978-0-904357-81-3.
- )
- .
- ^ ISBN 0-12-352651-5
- ISBN 978-0-08-037941-8.[page needed]
- ^ Taylor, F. Sherwood (1942). Inorganic & Theoretical Chemistry (6th ed.). Heineman.
- .
- .
- OCLC 64305455.
- ^
- S2CID 100214292. Archived from the original(PDF) on 2011-08-07. Retrieved 2009-07-31.
- S2CID 3011002.
- ISBN 978-0-935702-05-7.
- ^ Emsley 2001, pp. 124, 231, 449 and 503.
- ^ a b "Tin: From Ore to Ingot". International Tin Research Institute. 1991. Archived from the original on 2009-03-22. Retrieved 2009-03-21.
- ISBN 978-0-941375-62-7. Archivedfrom the original on 2016-04-28.
- ^ a b c d e f g Carlin, James F. Jr. "Tin: Statistics and Information" (PDF). United States Geological Survey. Archived from the original on 2008-12-06. Retrieved 2008-11-23.
- .
- ISBN 978-0-393-32831-8.
- .
- ^ "Seminole Group Colombia Discovers High Grade Tin Ore in the Amazon Jungle". FreePR101 PressRelease. Archived from the original on 2014-08-26.
- ISBN 978-0-87263-517-3. Archivedfrom the original on 2016-05-11.
- ^ Louis, Henry (1911). Metallurgy of tin. McGraw-Hill book Company.
- ISBN 978-0-8047-2136-3. Archivedfrom the original on 2016-05-19.
- ^ "LME Tin Brands". ITRI.co.uk. International Tin Research Institute. Archived from the original on 2008-12-07. Retrieved 2009-05-05.
- ^ "Top Ten Tin Producing Companies". itri.co.uk. International Tin Research Institute. Archived from the original on 2008-12-07. Retrieved 2009-05-05.
- ^ "The World's Biggest Tin Producers". The Balance. 13 January 2019.
- ^ "Agreement establishing the Association of Tin Producing Countries [1984] ATS 10". Australasian Legal Information Institute, Australian Treaties Library.
- ^ a b Carlin, James F. Jr. (1998). "Significant events affecting tin prices since 1958" (PDF). USGS. Archived from the original (PDF) on 2011-10-28.
- ^ "12 Januari Pemasaran Perdana INATIN". 15 December 2011. Archived from the original on 2012-04-26.
- ^ Daly, Tom (5 December 2021). "Tin surge worsens supply chain woes for electronics, solar and auto firms". Reuters. Retrieved 2021-12-07.
- ^ "Tin demand to decline – International Tin Association". Mining.com. 18 October 2019. Retrieved 2021-07-03.
- PMID 16203230.
- ^ "Technical data Sheet - Lead free alloy" (PDF). RS Online. Retrieved 2023-06-18.
- ^ Childs, Peter (July 1995). "The tin-man's tale". Education in Chemistry. Vol. 32, no. 4. Royal Society of Chemistry. p. 92. Retrieved 2018-06-19.
- ISBN 978-0-8047-2136-3. Archivedfrom the original on 2016-05-31.
- ^ Panel On Tin, National Research Council (U.S.). Committee on Technical Aspects of Critical and Strategic Materials (1970). Trends in the use of tin. pp. 10–22. Archived from the original on 2016-05-22.
- ISBN 978-0-7478-0152-8.
- ISBN 978-1-110-11092-6.
- ISBN 978-1-4067-4671-6.
- ^ "The Composition of the Cent". US Mint. Archived from the original on 2011-10-25. Retrieved 2011-10-28.
- ^ "Composition of canadian coins" (PDF). Canadian Mint. Archived (PDF) from the original on 2012-01-13. Retrieved 2011-10-28.
- doi:10.1063/1.881384.
- ISBN 978-0-87170-867-0. Archivedfrom the original on 2016-05-28.
- ISBN 978-0-415-94174-7.
- ISBN 978-0-486-21315-6.
- doi:10.1063/1.371708.
- ISBN 978-0-89134-739-2.
- ^ "Tin punching". Archived from the original on 2011-08-11. Retrieved 2011-08-15.
- S2CID 109981215.
- .
- ^ "Colgate Gel-Kam". Archived from the original on 2009-04-27. Retrieved 2009-05-05.
- PMID 2732364.
- PMID 8593194.
- ISBN 978-0-85404-459-7. Archivedfrom the original on 2016-05-21.
- ^ ISBN 978-0-7167-4878-6.
- ISBN 978-1-56990-379-7. Archivedfrom the original on 2016-05-09.
- ISBN 978-0-8247-0024-9.
- PMID 17959247.
- ^ Eisler, Ronald. "Tin Hazards To Fish, Wildlife, and Invertebrates: A Synoptic Review" (PDF). U.S. Fish and Wildlife Service Patuxent Wildlife Research Center. Archived (PDF) from the original on 2012-01-18.
- ^ "Regulation (EC) No 782/2003 of the European Parliament and of the Council of 14 April 2003 on the prohibition of organotin compounds on ships". europa.eu. Retrieved 2009-05-05.
- ISBN 978-1-4051-6926-4.
- ^ .
- .
- ^ PMID 12605923.
- ^ a b c "The Effects of Tributyltin on the Marine Environment". ScienceBuzz. 17 November 2018. Archived from the original on 2021-01-25. Retrieved 2020-11-17.
- ISBN 0-471-26418-0.
- PMID 32266205.
- .
- ^ "NIOSH Pocket Guide to Chemical Hazards – Tin". CDC. Archived from the original on 2015-11-25. Retrieved 2015-11-24.
Bibliography
 This article incorporates text from this source, which is in the public domain: Carlin, James F., Jr. (1998). "Significant events affecting tin prices since 1958". U.S. National Geodetic Survey
This article incorporates text from this source, which is in the public domain: Carlin, James F., Jr. (1998). "Significant events affecting tin prices since 1958". U.S. National Geodetic Survey- Lide, David R., ed. (2006). Handbook of Chemistry and Physics (87th ed.). Boca Raton, Florida: CRC Press, Taylor & Francis Group. ISBN 978-0-8493-0487-3.
- Emsley, John (2001). "Tin". Nature's Building Blocks: An A–Z Guide to the Elements. Oxford, England, UK: Oxford University Press. pp. 445–450. ISBN 978-0-19-850340-8.
- ISBN 978-0-08-037941-8.
- Heiserman, David L. (1992). "Element 50: Tin". Exploring Chemical Elements and their Compounds. New York: TAB Books. ISBN 978-0-8306-3018-9.
- MacIntosh, Robert M. (1968). "Tin". In Clifford A. Hampel (ed.). The Encyclopedia of the Chemical Elements. New York: Reinhold Book Corporation. pp. 722–732. LCCN 68-29938.
- Stwertka, Albert (1998). "Tin". Guide to the Elements (Revised ed.). Oxford University Press. ISBN 978-0-19-508083-4.
External links
- Tin at The Periodic Table of Videos(University of Nottingham)
- Theodore Gray's Wooden Periodic Table Table: Tin samples and castings
- Base Metals: Tin
- CDC – NIOSH Pocket Guide to Chemical Hazards
- Tin (USD cents per kg)
